Author: María Merino
A recent article published in Nature by Andrew L. Fanning and Kate Raworth (1) puts forward an uncomfortable but increasingly difficult-to-ignore idea: improving some social indicators is not enough if that progress comes at the expense of the ecological systems that sustain health and life.
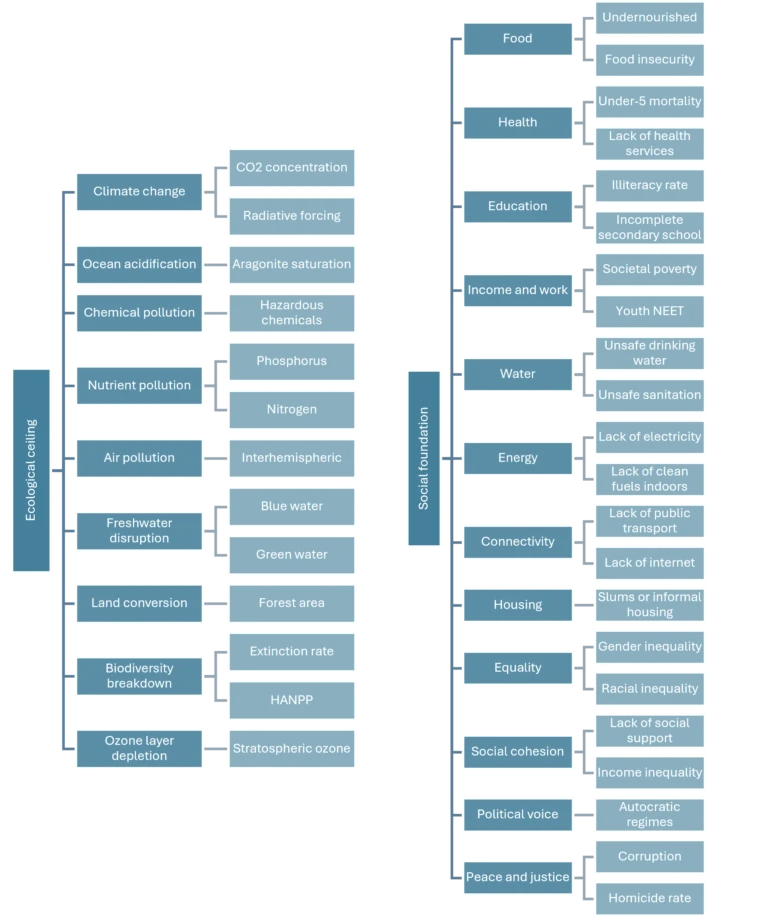
To demonstrate this, the authors update the Doughnut framework and build a global monitor with 21 dimensions and 35 indicators (figure 1), tracking over two decades (from 2000 to 2022) two sides of the same problem: social deprivation and the transgression of planetary ecological boundaries.
Figure 1. Dimensions and indicators of the social foundation and the ecological ceiling.
HANPP: human appropriation of net primary productivity; NEET: youth not in employment, education or training.
Source: author’s own elaboration based on Andrew L. Fanning and Kate Raworth, 2025 (1).
The central message of the paper is clear. Although global GDP doubled over those two decades, the reduction in social deprivation was modest and, at the current pace, would need to accelerate fivefold in order to meet the basic needs of the entire population by 2030. Meanwhile, ecological overshoot has not only failed to improve, but has continued to worsen: the authors conclude that humanity has already exceeded at least six of the nine planetary boundaries.
The value of the article lies not only in its diagnosis, but also in the shift in perspective it proposes. Rather than viewing progress almost exclusively through the lens of economic growth, the Doughnut asks a more demanding question: are we meeting essential human needs within safe material and environmental limits? From that perspective, the current answer is “not yet”. And this has a very direct relevance for health technology assessment (HTA).
For years, HTA has mainly rested on three major pillars, which remain essential: clinical effectiveness, economic efficiency, and budget impact. However, the article suggests that this approach may be too narrow if it does not incorporate a broader perspective on sustainability, equity, and externalities. A technology may be clinically valuable and even cost-effective in QALY terms, while at the same time generating a high environmental footprint, depending on fragile supply chains, or producing highly unequal benefits across population groups. That is the conversation the article invites us to have.
This reflection is particularly relevant in the current Spanish regulatory context. The draft Royal Decree on Health Technology Assessment (2) does not define HTA merely as a clinical appraisal. Instead, it describes it as a multidisciplinary process that comparatively evaluates the medical, social, patient-related, economic, and ethical aspects of a health technology. The text also makes clear that HTA should inform decisions on adoption, funding, pricing, reimbursement, or disinvestment, and that its scope includes not only medicines, but also medical devices, in vitro diagnostics, procedures, digital technologies, and organisational models.
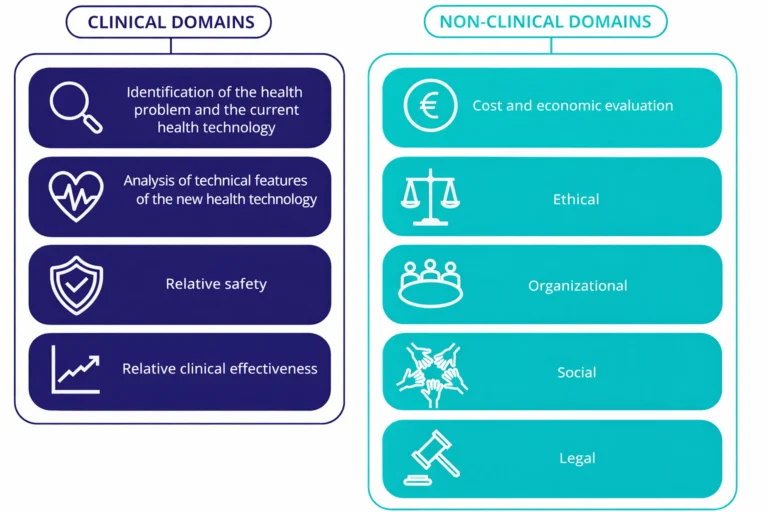
This is where the aforementioned article and the future Royal Decree converge. If the new Spanish framework genuinely intends to take these “non-clinical” dimensions seriously, the HTA of the future will need to look beyond the classic effectiveness-cost binary. The draft already points in that direction by separating clinical assessment from the analysis of other non-clinical aspects, including economic evaluation, and by explicitly introducing the use of real-world data and real-world evidence to support assessments and decision-making (figure 2).
Figure 2. Assessment domains included in the draft Royal Decree regulating Health Technology Assessment.
Source: author’s own elaboration based on González Domínguez et al., 2026 (3).
In addition, the Ministry has stressed that the new model incorporates the participation of patients and healthcare professionals in assessment processes. This is no minor detail. It means recognising that the value of a technology is not exhausted by traditional clinical outcomes, but also includes issues such as patient experience, treatment burden, acceptability, service organisation, or inequalities in access.
In parallel, Regulation (EU) 2021/2282 (4) already marked an important shift by reminding us that HTA may cover not only clinical aspects, but also non-clinical domains. At the European level, nine assessment domains are mentioned, four of them clinical and five non-clinical. Spain appears to be aligning itself with this evolution, but it could go even further if it uses this reform to integrate more systematically emerging dimensions such as environmental sustainability or the distributive impact of decisions.
Therefore, the main takeaway is that the value of a health technology can no longer be understood solely within the healthcare system, but must also be considered in relation to the social and ecological systems in which it operates. This is not about replacing traditional economic analysis or abandoning QALYs, but about complementing them. The question is no longer only “how much health benefit does this technology produce per euro invested?”, but also “what resources does it consume, what externalities does it generate, who truly benefits from it, and is that value sustainable over time?”.
There is no real health innovation if clinical progress is built on invisible social or ecological costs.
References
1. Fanning AL, Raworth K. Doughnut of social and planetary boundaries monitors a world out of balance. Nature. 2025;646: 47–56. https://doi.org/10.1038/s41586-025-09385-1.
2. Real Decreto XXXXXXX/2024, de X de XXXXXX, por el que se regula la evaluación de tecnologías sanitarias. https://www.sanidad.gob.es/normativa/audiencia/docs/DG_54_24_Solicitud_informacion_publica_RD_EVALUACION_TECNOLOGIAS_SANITARIAS.pdf [Accessed 11th June 2025].
3. González Domínguez A, Merino Ventosa M, Durán Esteban A, Comité técnico, Comité consultivo. Reflexión Estratégica sobre el VALOR social en la evaluación de medicamentos. 2026. https://doi.org/10.65616/OMEN2105.
4. Parlamento Europeo y Consejo de la Unión Europea. Reglamento (UE) 2021/2282 del Parlamento Europeo y del Consejo de 15 de diciembre de 2021 sobre evaluación de las tecnologías sanitarias y por el que se modifica la Directiva 2011/24/UE. Diario Oficial de la Unión Europea (OJ L), 2021/2282 2021. http://data.europa.eu/eli/reg/2021/2282/oj/spa [Accessed 12th June 2025].