Author: Julia Alonso
What began as a scientific promise has today become a clinical revolution thanks to the development of CAR-T cells (chimeric antigen receptor T cells). However, beyond their clinical impact, these therapies pose significant economic and equity challenges: Are they sustainable for public budgets? Do all patients have the same access opportunities?
What are CAR-T cells?
CAR-T therapy is an advanced immunotherapy procedure in which a patient’s own T lymphocytes are collected and genetically modified to recognize and destroy tumor cells and then re-administered to the patient (a procedure scientifically known as apheresis). CAR-T therapies have demonstrated highly relevant results in leukemias and lymphomas, particularly in patients with poor prognosis and limited therapeutic options (1,2). Overall survival is approximately 78% at 6 months and 52% at 18 months. However, these outcomes depend on the type of cancer treated, the target antigen, and the specific CAR-T product, as well as on patient characteristics (3).
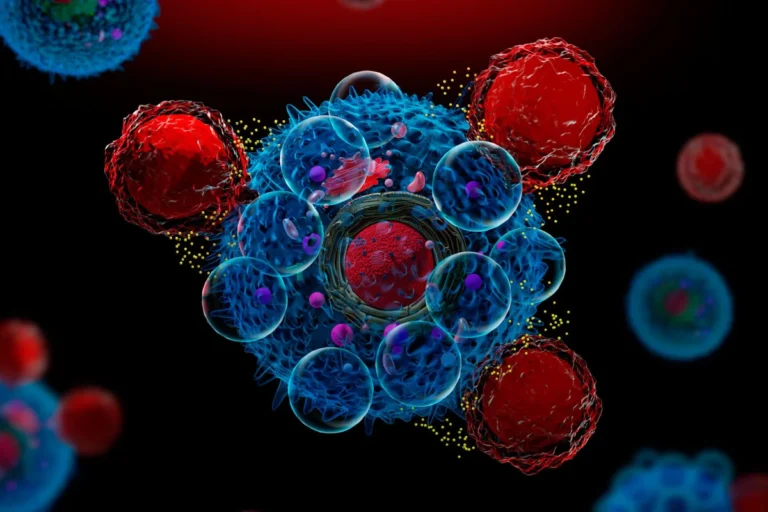
Figure 1. CAR-T cell attacking a cancer cell.
Reference: El Mundo (4).
As expected, innovation comes at a price. In Spain, the commercially available CAR-T therapies (Kymriah®, Yescarta®, Tecartus®, Abecma®, or Carvykti®) can exceed €300,000 per patient and treatment, not including costs associated with hospitalization, intensive care, and management of adverse effects (5).
In response to this high economic impact, the incorporation of CAR-T therapies into the healthcare system has driven the adoption of innovative financing models, such as payment-by-results agreements, aimed at sharing risks and limiting budgetary pressure (6). Additionally, they require real-world clinical monitoring systems (e.g., VALTERMED) to track health outcomes and ensure efficient resource use (7).
Academic CAR-T: a public alternative
Innovative responses to the high cost of commercial CAR-T therapies have emerged from the Spanish public healthcare system. While industrially manufactured CAR-T therapies authorized by the European Commission are developed by large pharmaceutical companies on a large scale and under standardized processes, undergoing rigorous clinical trials and enabling uniform and accessible treatments for the approved indications, academic CAR-T therapies originate from university hospitals and public centers, often under the hospital exemption clause, allowing for greater customization, targeting different antigens, with shorter development times and lower costs. (8). The Hospital Clínic de Barcelona has developed academic CAR-T therapies, such as ARI-0001 and ARI-0002h, already approved by the Spanish Agency of Medicines and Medical Devices (AEMPS), while other hospitals are working on new proposals. These therapies significantly reduce manufacturing and financing costs—in the case of ARI-0001, the list price is €89,000 per patient—and open the door to more sustainable and equitable models (5,9).
Unequal access between autonomous communities
Despite these advances, access to CAR-T therapies remains unequal in Spain. Although most regions have authorized centers, others, such as Castilla-La Mancha, Extremadura, or La Rioja, do not, requiring patients to be referred to other regions (10). However, this referral does not always guarantee effective access to treatment (11). Travel, waiting times, administrative complexity, and associated indirect costs can become real barriers that limit therapeutic options, raising questions about whether a patient’s place of residence effectively determines the possibility of receiving a potentially curative therapy.
Final reflection on the challenges and opportunities of CAR-T therapies
CAR-T therapies represent a revolutionary leap in medicine, offering hope where few options previously existed. Science continues to advance, with Tandem CAR-T therapies under development and the potential for outpatient CAR-T treatments being considered (12). Undoubtedly, the main challenges remain in research and development, price and reimbursement models, investment in infrastructure, training, and establishing an appropriate regulatory framework. Yet all of this constitutes a tremendous opportunity: the chance to save lives.
It is worth closing this article with a final reflection on the social value generated by CAR-T therapies. If we were to consider savings from avoided losses in labor productivity of patients and their caregivers, quality-adjusted life years (QALYs) gained, and premature deaths prevented due to treatment, the resulting social impact would likely more than justify the investment.
Referencias
1. Zhang X, Zhu L, Zhang H, Chen S, Xiao Y. CAR-T Cell Therapy in Hematological Malignancies: Current Opportunities and Challenges. Front Immunol. 2022;13:927153.
2. Bharadia H, Dabhade A, Shah AC, Patel R, Chorawala MR, Patel A, et al. CAR T-cell immunotherapy as the next horizon in cancer eradication: current landscape, challenges, and future directions. Med Oncol. 6 de agosto de 2025;42(9):410.
3. Wang V, Gauthier M, Decot V, Reppel L, Bensoussan D. Systematic Review on CAR-T Cell Clinical Trials Up to 2022: Academic Center Input. Cancers. enero de 2023;15(4):1003.
4. Barcelona MR. ELMUNDO. 2023 [citado 16 de diciembre de 2025]. Terapias CAR-T y tumores sólidos: primeros resultados positivos. Disponible en: https://www.elmundo.es/ciencia-y-salud/salud/2023/12/02/656a0a4efdddff06288b4572.html
5. www.ocu.org [Internet]. [citado 16 de diciembre de 2025]. Terapias oncológicas avanzadas : la primera CAR-T pública. Disponible en: https://www.ocu.org/salud/medicamentos/noticias/terapia-cart-publica
6. Ministerio de Sanidad. Ministerio de Sanidad – Profesionales de la Salud – Buscador situación financiación medicamentos [Internet]. [citado 17 de diciembre de 2025]. Disponible en: https://www.sanidad.gob.es/profesionales/medicamentos.do
7. Ministerio de Sanidad. Ministerio de Sanidad – Profesionales – VALTERMED: Resultados en salud [Internet]. [citado 17 de diciembre de 2025]. Disponible en: https://www.sanidad.gob.es/areas/farmacia/infoMedicamentos/valtermed/
8. Grupo Español de Transplante Hematopoyético y Terapia Celular. CART [Internet]. 2020 [citado 17 de diciembre de 2025]. Disponible en: https://www.geth.es/component/content/article/226-cart-cells/609-cart-industrial-y-academico
9. Delgado J, Juan M, Calvo G, Urbano-Ispizua Á. The development of academic CAR T cells. The Lancet Haematology. 2024;11(7):e484-5.
10. Ministerio de Sanidad. Ministerio de Sanidad – Áreas – Terapias Avanzadas en el Sistema Nacional de Salud (SNS) [Internet]. [citado 16 de diciembre de 2025]. Disponible en: https://www.sanidad.gob.es/areas/farmacia/infoMedicamentos/terapiasAvanzadas/home.htm?utm_source=copilot.com
11. SEHH (Sociedad Española de Hematología y Hemoterapia). Guía IDEaL. Guía de recomendaciones para la identificación y derivación temprana de pacientes con linfoma no Hodgkin de células B candidatos a terapias CAR-T. 2024.
12. Arganda C. CAR-T ambulatorio: misma eficacia y seguridad, con más eficiencia y calidad de vida | @diariofarma [Internet]. diariofarma. 2025 [citado 30 de diciembre de 2025]. Disponible en: https://diariofarma.com/2025/10/17/car-t-ambulatorio-misma-eficacia-y-seguridad-con-mas-eficiencia-y-calidad-de-vida